Guideline Consensus | Hiatal Hernia Repair and Antireflux Surgery: Standard Micro-Consensus
Gastroesophageal reflux disease (GERD) is a common digestive system disease with an increasing incidence year by year [1]. In 2006, 2007, and 2014, China issued guidelines for the medical diagnosis and treatment of GERD [2-4], and in 2020, the Chinese expert consensus on gastroesophageal reflux disease was published, which has played an important role in guiding the clinical management of GERD. In recent years, due to the application of surgical intervention in the clinical practice and research of GERD, there is an urgent need for a domestic surgical consensus to standardize treatment, provide a clinical framework for clinicians in this field, and guide physicians in related disciplines to correctly understand and manage this disease. The recommendations presented in this consensus are derived from a systematic review of published literature on GERD and evidence-based clinical recommendations. The strength of the evidence is highlighted, and expert opinion was adopted in areas lacking conclusive evidence.
I. Definition and Symptoms
GERD is defined as symptoms and complications resulting from the reflux of gastric contents into the esophagus, oral cavity (including the larynx), or lungs [5]. Typical symptoms of GERD are heartburn and regurgitation. Heartburn refers to a retrosternal burning sensation, and regurgitation refers to the sensation of gastric contents flowing toward the pharynx or oral cavity. Atypical symptoms include chest pain, upper abdominal pain, epigastric burning sensation, and belching. GERD is also associated with extraesophageal symptoms, including cough, laryngeal symptoms, asthma, and dental erosion. In patients with concomitant hiatal hernia, some may present with anemia.
A hiatal hernia is defined as the herniation of abdominal viscera through the esophageal hiatus of the diaphragm. Most small type I (sliding) hiatal hernias are asymptomatic. Patients with larger type I hiatal hernias may present with symptoms of GERD, most commonly heartburn, regurgitation, and dysphagia. Many patients with type II, III, and IV (paraesophageal) hernias are asymptomatic or have only vague, intermittent symptoms. The most common symptoms are epigastric or retrosternal pain, postprandial fullness, nausea, and retching. Compared with patients with type I hiatal hernia, GERD symptoms are less common in these patients.
II. Classification
Clinically, GERD is generally classified into three types: erosive esophagitis (EE) or reflux esophagitis (RE), non-erosive reflux disease (NERD), and Barrett’s esophagus (BE) [7]. Hiatal hernias can be broadly divided into sliding hernias and paraesophageal hernias. The most general classification of hiatal hernias includes four types:
Type I: Sliding hernia – Type I or sliding hiatal hernia is characterized by displacement of the gastroesophageal junction above the diaphragm. The stomach retains its normal longitudinal orientation, and the gastric fundus remains below the gastroesophageal junction.
Types II, III, and IV: Paraesophageal hernia – A paraesophageal hernia is a true hernia with a hernia sac, characterized by the upward herniation of the gastric fundus through a defect in the phrenoesophageal membrane.
Type II hiatal hernia results from a localized defect in the phrenoesophageal membrane, with the gastric fundus serving as the leading point of herniation, while the gastroesophageal junction remains fixed to the preaortic fascia and the median arcuate ligament.
Type III hiatal hernia shares features of both type I and type II hiatal hernias, characterized by herniation of both the gastroesophageal junction and the gastric fundus through the esophageal hiatus. The gastric fundus is located above the gastroesophageal junction.
Type IV hiatal hernia is associated with a large defect in the phrenoesophageal membrane and is characterized by the presence of other organs (such as the colon, spleen, pancreas, or small intestine) within the hernia sac in addition to the stomach [7].
III. Diagnosis
Diagnostic and treatment criteria: (1) presence of typical symptoms such as acid regurgitation, heartburn, and retrosternal pain; (2) positive proton pump inhibitor test; (3) endoscopy showing positive for reflux esophagitis; (4) positive 24-hour esophageal pH monitoring; (5) GERD-Q score: the questionnaire assesses the frequency of six symptoms (heartburn, regurgitation, upper abdominal pain, nausea, sleep disturbance, and need for over-the-counter medication) over one week, with a total score of 18; a score ≥8 is diagnostic of GERD [8]; (6) high-resolution esophageal manometry (HRM) can simultaneously monitor the tone and peristaltic contractions of the entire esophagus. It helps to determine the presence of a hiatal hernia, reflects esophageal motor function, and provides a basis for selecting the surgical approach [9]. Paraesophageal hernia may be diagnosed by upper gastrointestinal endoscopy, but barium swallow esophagography is the most sensitive test for diagnosis. A sliding hiatal hernia >2 cm can be diagnosed by barium swallow, endoscopy, or esophageal manometry. In contrast, smaller sliding hiatal hernias can only be diagnosed intraoperatively. CT is not routinely recommended but may provide additional information regarding the location and type of hiatal hernia. Typically, it is found incidentally on CT scans performed for different indications [10].
IV. Treatment Strategies
The treatment goals for GERD include effective symptom relief, healing of esophagitis, improvement of quality of life, and prevention of complications. We strongly recommend that asymptomatic type I hiatal hernia does not require surgical repair. Patients with symptomatic sliding hiatal hernia require concurrent priority treatment of GERD. The optimal management of asymptomatic paraesophageal hernia is controversial. While some experts recommend prophylactic surgical treatment even in asymptomatic patients, others advise against it. Symptomatic or complicated paraesophageal hernias (types II, III, and IV) require surgical repair; the urgency of surgery depends on the rapidity of onset.
Indications for emergency repair include acute gastric volvulus, uncontrolled bleeding, obstruction, strangulation, perforation, or respiratory impairment secondary to paraesophageal hernia. Paraesophageal hernia causing acute symptoms carries a high mortality rate.
Indications for elective repair include subacute symptoms in the setting of paraesophageal hernia, such as dysphagia, early satiety, postprandial chest or abdominal pain, anemia, vomiting, or GERD refractory to medical management. Surgical repair improves symptoms and quality of life (QOL) in these patients [10-11].
V. Surgical Techniques and Key Points
1. Standardization of surgical technique
Standardized anti-reflux surgery can improve patient outcomes. Like any surgical procedure, laparoscopic anti-reflux surgery is subject to a learning curve, which may affect treatment results. Therefore, surgeons who are not proficient in advanced laparoscopic procedures, especially fundoplication, must perform their initial laparoscopic cases under expert guidance to reduce complication rates and improve outcomes. Repeat anti-reflux surgery should be performed by experienced surgeons at large tertiary medical centers.
2. Division of short gastric vessels
According to evidence from five randomized controlled trials with follow-up of up to 10 years, there were no differences in surgical outcomes such as physiology, symptomatology, and quality of life. Moreover, division of short gastric vessels during fundoplication prolongs operative time, increases the incidence of bloating and epigastric gas, and impairs gastric排气 function. It is recommended to preserve the short gastric vessels when the fundus can be wrapped without tension. Division of the short gastric vessels should be performed when the fundus cannot be wrapped without tension.
3. Preservation of the vagus nerve
Vagus nerve injury is a relatively common complication during anti-reflux surgery. Although there is no difference in short-term outcomes such as diarrhea, satiety, dysphagia, and regurgitation between patients with and without vagus nerve injury, long-term management of reflux symptoms is associated with higher recurrence and reoperation rates. Therefore, we believe that surgeons should operate carefully to preserve the integrity of the vagus nerve whenever possible, especially in patients with gastroesophageal reflux disease. During opening of the lesser omental sac and dissection of the right diaphragmatic crus, attention should be paid to preserving the hepatic branch of the vagus nerve to avoid increasing the risk of cholelithiasis.
4. Closure of the esophageal hiatus
During fundoplication, closure of the esophageal hiatus (suture of the crura) should be considered when the hiatal defect is large. Mesh reinforcement may help reduce the incidence of wrap herniation. Anterior closure of the esophageal hiatus may reduce the incidence of postoperative dysphagia, but more evidence is needed to support this recommendation. Any hiatal hernia found during Roux-en-Y gastric bypass, sleeve gastrectomy, or adjustable gastric band placement should be repaired. Non-absorbable monofilament polypropylene suture is recommended for cruroplasty.
5. Use of mesh
Indications for mesh use: (1) giant hiatal hernia with a hiatal defect >5 cm or herniation of more than one-third of the gastric body into the chest; (2) patients with poorly developed crura and thin muscle fibers, where simple suture repair is prone to tearing of the crura and diaphragmatic muscle fibers with respiratory and swallowing movements, leading to recurrence of hiatal hernia; (3) patients with concomitant chronic diseases (obesity, constipation, benign prostatic hyperplasia, chronic bronchitis, intractable hiccups, etc.) that increase intra-abdominal pressure, forcing abdominal viscera toward the esophageal hiatus and into the chest through the hiatus [15].
6. Choice of anti-reflux surgical procedure
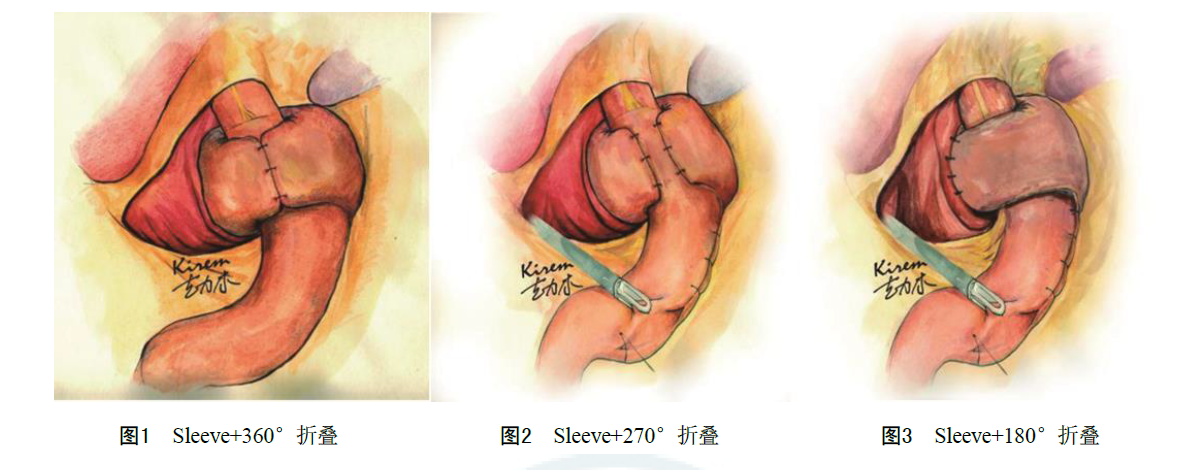
Laparoscopic anti-reflux procedures mainly include Nissen (360°) fundoplication, Toupet (270°) fundoplication, and Dor (180°) fundoplication. See Figures 1–3. According to available high-quality evidence, compared with complete fundoplication, partial fundoplication is associated with lower rates of postoperative dysphagia and reoperation at 5 years, with comparable patient satisfaction and GERD control. Most literature suggests that the long-term outcomes of anterior fundoplication are not ideal, and retrospective data show that the long-term outcomes of partial fundoplication are inferior to those of complete fundoplication. However, available evidence suggests that when GERD surgery is performed by appropriately trained minimally invasive surgeons, and with the use of an esophageal bougie, partial fundoplication or a short (1–2 cm) complete fundoplication may minimize postoperative dysphagia; complete fundoplication or a longer (at least 3 cm) posterior fundoplication may achieve the best treatment outcomes.
VI. Anti-reflux Surgery in Obese Patients
Due to concerns about higher failure rates of fundoplication in morbidly obese patients (body mass index >35 kg/m²) and the inability of fundoplication to address underlying obesity and its related comorbidities, bariatric surgery should be considered concurrently in obese patients preparing for anti-reflux surgery. The preferred approach is Roux-en-Y gastric bypass, or sleeve gastrectomy should be performed in combination, or gastric bypass should be considered directly [16].
VII. Robot-Assisted Laparoscopic Anti-reflux Surgery
Although robot assistance can be used safely and effectively for fundoplication, its higher cost and similar short-term patient outcomes compared to conventional laparoscopy make it not an ideal initial choice. However, further studies on the learning curve and surgeon workload of robotic technology are needed before stronger recommendations can be made [17].

