Gynecology & Urology
Partially absorbable urethral sling
Indications
For use in mid-urethral sling surgery in female patients with stress urinary incontinence caused by excessive urethral mobility and/or sphincter dysfunction.
Composition
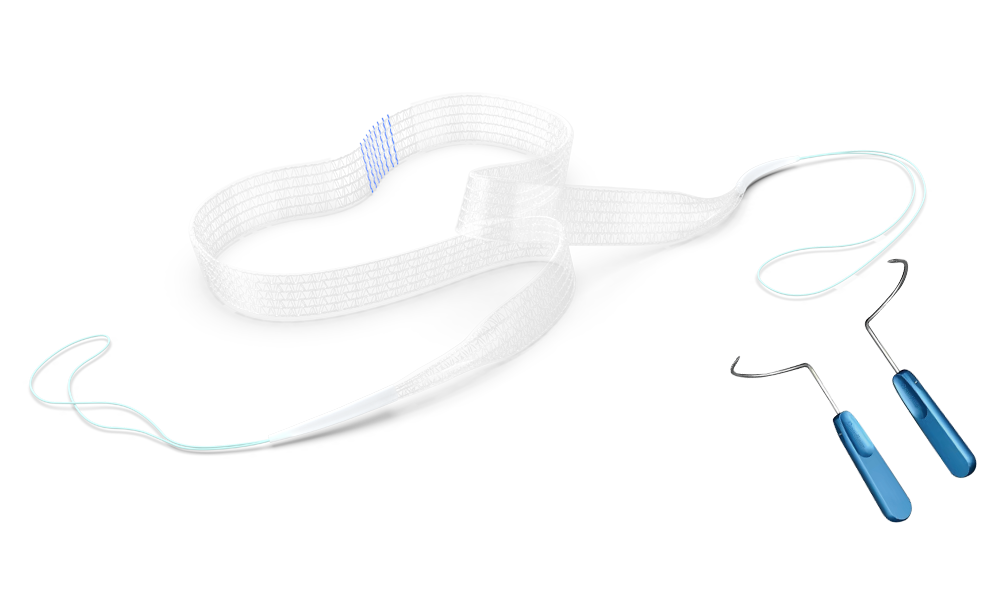
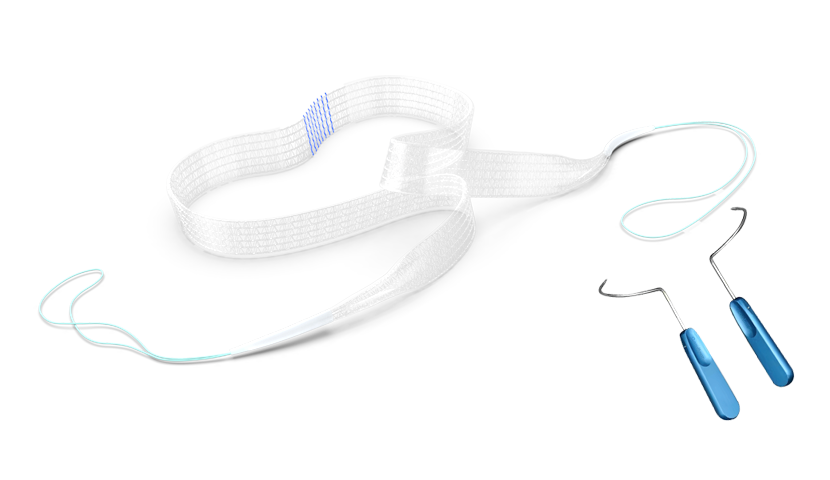
The product consists of a sling and an instrument. The sling comprises a mesh and a connecting end, while the instrument is composed of a 304 stainless steel body and a handle. The mesh is co-woven from polypropylene monofilament and absorbable poly(L-lactide) monofilament. One side of the mesh is evenly distributed with absorbable hooks made of poly(L-lactide) monofilament, and the positioning thread is dyed with phthalocyanine blue.
Product Features
| Easy-to-use pre-loaded design: | Distinct blue midline marker for easy identification, facilitating precise intraoperative positioning and better handling.
Pre-loaded protective sheath enables smoother retraction within the body and reduces trauma and irritation to surrounding tissues. |
| World’s first self-fixing urinary incontinence sling design: | Poly(L-lactide) micro-hook design – self-fixing micro-hooks prevent migration, ensuring clinical efficacy.
“Mushroom-head” micro-hook with a blunt, rounded tip embeds into tissue, minimizing tissue damage. |
| Unique weaving technology: | Proprietary mesh structure maintains a constant effective area under tension, extremely low shrinkage rate, and fewer postoperative complications.
High effective pore ratio, soft and easily deployable mesh, excellent tissue conformability. |
| Optimally dense absorbable micro-hooks: | Evenly distributed micro-hooks provide uniform force distribution and greater tensile strength.
Absorbable micro-hooks fully degrade after 18 months, reducing total graft weight by 50% and leaving less foreign material. |
Related Products
Contact Us
Interested in our products? Please fill in your information and we will contact you as soon as possible.